Displaying items by tag: control
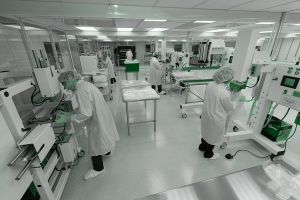
Cleanroom – Definition, Purposes And Equipment Of Controlled Environments
Several terms are currently used to describe controlled environments, but they are most often referred to as clean zones, or just ‘cleanrooms’. The key role of this type of rooms is to ensure that the air there has certain precisely specified parameters that are necessary to ensure the repeatability of production, regardless of the industry, be it pharmaceutics, electronics or the automotive industry.
Introduction to Clean Room Technology and Contamination Control
Clean room technology plays a vital role in various industries, including pharmaceuticals, biotechnology, medical devices, electronics, and aerospace, where the control of contamination is crucial. Clean rooms are controlled environments designed to minimize the presence of airborne particles, microorganisms, and other contaminants that could impact the quality of products or processes. Maintaining a clean room environment is essential to ensure the integrity, safety, and reliability of the products being manufactured.
Cleanroom Best Practices: 18 Rules For Cleaner Controlled Environments
Check out our list of Dos and Don’ts in a cleanroom, a summary of cleanroom best practices for making the controlled environment function at peak performance. These are good resources for new cleanroom operators or those unsure about what steps to take in a cleanroom environment.
The Basics Of Cleanroom Design & Material Transfer For Microbial Control
Microbial control is critical in cleanroom environments. Contaminated environments can lead to product recalls, regulatory observations, fines, or even consumer deaths. In order to properly prevent, destroy, and monitor microbial contamination in cleanrooms, several aspects of cleanroom microbiology must be understood. This foundational introduction to cleanroom microbiology discusses some of those aspects.
Cleanroom Control and Maintenance
Proper cleanroom cleaning procedure and maintenance protocol is an immediate, low-cost measure to enhance overall cleanliness, consistency, and contamination control within cleanrooms. This guide provides a framework for cleanroom management, protocol standards, specifications, and processes for general manufacturing and biomedical applications. Because each cleanroom class can require slightly different supplies and protocol, this overview connects best practices with application specific techniques for gowning, wipedown, and particle control. This guide should serve as a resource for understanding broad-line cleanroom consumable needs, but not as a replacement for expertise tailored to your application.
Humidity Control for Clean Rooms
Clean Rooms are workplaces where contamination is controlled. The Clean Rooms are said to provide a conducive environment (for research, development and manufacturing of equipment or processes) with a low level of environmental pollutants such as dust, airborne microbes, aerosol particles, and chemical vapors.
Cleanroom Particle Control
Cleanrooms are designed to minimise the ingress of airborne particles (achieved through HEPA or ULPA filters) and to control what happens to particles generated within the cleanroom. Good air flow design — such as turbulent flow — helps to prevent particles from being deposited onto surfaces (particles settle by two primary mechanisms: gravitational sedimentation and turbulent deposition)1. The removal of these particles is achieved through the extraction of room air with the addition of clean air into the room (air exchange rates). The flow of particles in air from a less clean area can also be blocked from entering an area of a higher cleanliness level through positive pressure differentials.
What Makes a Clean Room “Clean”?
Clean rooms are “created” when clean room designers like Vernick & Associates bring together engineering design, fabrication, finish, and operational controls to convert a “normal” room to a “clean room” so that they can be used for manufacturing. These clean rooms must meet the requirements defined in the Sterile Code of Annex 1 of both the EU and PIC/S Guides to GMP and other standards and guidance as required by local health authorities.
Cleanliness Control
As with cleanroom operations, cleanliness is a four-part equation: environment, process, tools, and people. How stringent clean manufacturing guidelines need to be is based on the products and customer requirements.
Controlling air temperature and humidity levels in all work areas is a good first step in clean manufacturing. It may eliminate the need to open windows, which would admit outdoor airborne dirt.
Five stages control at construction
As the demand for complex cleanrooms in the life sciences grows, construction challenges mount. Making the right decisions during the building and commissioning phases of cleanrooms will help to ensure that they are successful, better integrated, more operable and sustainable. Generally speaking, teamwork among designers, operators, maintenance people and builders that focuses on project timelines will achieve the best results. The suggestions below will help drug and device manufacturers step back and better envision the whole facility during the build process.